Ayar-Kayali Lab. on Biopharmaceutic Technologies and Bioanalysis
OVERVIEW
Our studies till today, we have continued to clarify the PKC signal pathway, the results from these studies will guide our new projects. We found that EpCAM, Claudin and tetraspanin are the important proteins that have been found effective in the signaling pathway.
Following this project, studies on the effect of exosomes released from ovarian cancer cells by primary ovarian cancer cells and healthy epithelium and mesothelial cells by uptake of 1001 Tubitak Project and carcinogenesis mechanism in target cells are continuing.
According to the results obtained from these studies, it is aimed to improve the diagnosis and treatment of ovarian and colon cancer. In this context, synthesis of targeted therapeutic molecules, development of diagnostic kits optimized for diseases and cancer, and full characterization of synthesized molecules are studied.
RESEARCH INTERESTS
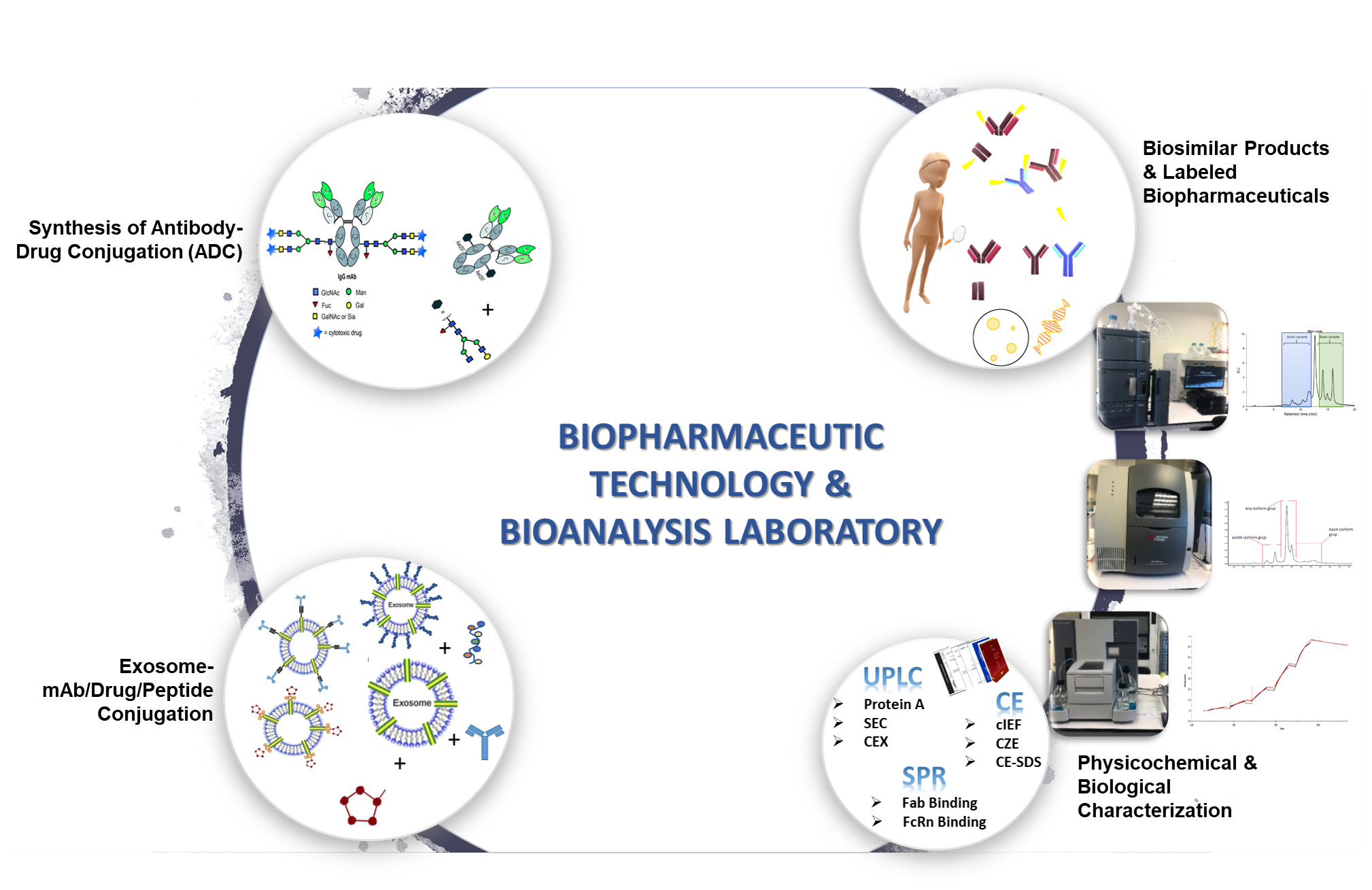
1. Development of the Smart (Targeted) Chemotherapeutic Agents and Drug Delivery System
The selective delivery of highly effective drugs by antibody drug conjugates (ADC) is a promising approach for the treatment of cancer to overcome the multidrug resistance (MDR). The monoclonal antibody (mAb), which has been highly expressed by specific cancer type, may be linked to payload which targets the DNA or microtubules using the site-specific conjugation methods to synthesize the ADC. Both the use of the target antibody and the use of a hydrophilic linker may help to overcome the MDR. In addition, we are planning to produce surface functionalized exosome (mAb-exosome, drug-exosome, peptide-exosome conjugates) loaded drug for specific targeting.
2. Development of the Diagnosis Kits
In this content, it is tried to find specific biomarkers for rare diseases and cancer. The biological nanovesicles, namely exosomes which include some specific proteins called “Exosome Markers”. The study aims to develop diagnostic kit based on exosome to help diagnose cancer types at an early stage and also determine the stage of the cancer by using statistical algorithms. The specific biomarker in the patient’s blood samples will be screened by using Exosome-mAb/Drug/Peptide Conjugation.
Group Members

Ayar-Kayali Lab. on Biopharmaceutic Technologies and Bioanalysis
Research Group Leader
Hülya AYAR KAYALI
hulya.kayali@ibg.edu.tr
+90 232 299 41 00
(5081)
+9
0 232 299 41 58

Gizem YILMAZER ALTUN
R&D Personnel
gizem.yilmazer@ibg.edu.tr

Mariam J. M GHUNAIM
PhD Student
mariam.ghunaim@ibg.edu.tr

Ekrem TINAZ
Researcher
ekrem.tinaz@ibg.edu.tr

Ege Gökçe SAVAŞ
PhD Student
ege.savas@ibg.edu.tr

Aytaj ABUSHOVA
PhD Student
aytaj.abushova@ibg.edu.tr

Elçin ÇAĞATAY
PhD Student
elcin.cagatay@ibg.edu.tr

Ezgi İTİL
PhD Student
ezgi.itil@ibg.edu.tr

Defne GÖVEM
MSc Student
defne.govem@ibg.edu.tr

Cengiz Han YAVUZ
Undergraduate Student
cengiz.yavuz@ibg.edu.tr

Zeynep ÖNCÜ
Undergraduate Student
zeynep.oncu@ibg.edu.tr

Duygu ERDOĞAN
Post-Doc Researcher
duygu.erdogan@ibg.edu.tr

Yonca GÜNGÖR
PhD Student
yonca.gungor@ibg.edu.tr

Onur ÖZKAYA
MSc Student
onur.ozkaya@ibg.edu.tr

Hilal TAY
MSc Student
hilal.tay@ibg.edu.tr

Halit Enes BELGE
MSc Student
halit.belge@std.ibg.edu.tr
Former Members

Seminay GÜLER
PhD Student
seminay.guler@ibg.edu.tr

Gülnur ÇIRAK
MSc Student
gulnur.cirak@msfr.ibg.edu.tr

Zehra TAVŞAN
Post-Doc Researcher
zehra.tavsan@msfr.ibg.edu.tr

Gizem KURŞUNOĞLU
Researcher
gizem.kursunluoglu@ibg.edu.tr

Elçin ÇAĞATAY
PhD Student
elcin.cagatay@ibg.edu.tr

Gizem YILMAZ
MSc Student
gizem.yilmaz@msfr.ibg.edu.tr

Egemen Erdem GÜLER
PhD Student
egemen.guler@ibg.edu.tr

Esra BULUT
Visiting Researcher
esra.bulut@ibg.edu.tr

Aziz Umut DURAK
MSc Student
umut.durak@ibg.edu.tr

Aziz Umut DURAK
MSc Student
umut.durak@ibg.edu.tr

Yonca GÜNGÖR
PhD Student
yonca.gungor@ibg.edu.tr

Ayşe CAŞKIR
MSc Student
ayse.caskir@ibg.edu.tr

Yiğit KOZALI
MSc Student
yigit.kozali@ibg.edu.tr

Buse SEVGİ
MSc Student
buse.sevgi@ibg.edu.tr

Duygu ERDOĞAN
PhD Student
duygu.erdogan@ibg.edu.tr

İlyas Umur AYAZ
Researcher
umur.ayaz@ibg.edu.tr

Enes GÜNDÜZ
Undergraduate Student
enes.gunduz@ibg.edu.tr

Elçin ÇAĞATAY
Researcher
elcin.cagatay@ibg.edu.tr

Muzaffer DÜKEL
Visiting Researcher
muzaffer.dukel@ibg.edu.tr

Hezzal KÜÇÜKSELBES
Undergraduate Student
hezzal.kucukselbes@ibg.edu.tr

Ekrem TINAZ
Visiting Researcher
ekrem.tinaz@ibg.edu.tr

PUMLA BHEKIWE MANYATSI
PhD Student
pumlabhekiwe.manyatsi@ibg.edu.tr

Fethican KAYHAN
MSc Student
fethican.kayhan@ibg.edu.tr

Gamze ÜNER
Visiting Student
None

Gaye SAYGI
Visiting Researcher
gaye.saygi@ibg.edu.tr

İlayda KELEŞ
MSc Student
ilayda.keles@ibg.edu.tr

Oğuzhan AVCI
Undergraduate Student
oguzhan.avci@ibg.edu.tr

Duygu ERDOĞAN
PhD Student
duygu.erdogan@ibg.edu.tr

Ece ÖNEL
Undergraduate Student
ece.onel@std.ibg.edu.tr
Projects
The Scientific and Technological Research Council of Turkey - TUBITAK - RD : Ovaryum Kanserinde Tetraspain-Claudin-Epcam Tümör Belirleyici Proteinlerinin ve Protein Kinaz C'nin Rolünün Belirlenmesi, Finished
The Scientific and Technological Research Council of Turkey - TUBITAK - RD : Kanser ve Osteoporoz Tedavisi İçin Monoklonal Antikor Etkin Maddeli Biyobenzer İlaç Geliştirilmesi ve Üretilmesi, Ongoing
DEÜ BAP - Dokuz Eylul University Scientific Research Projects Coordination Unit - RD : Kolon Kanserinde Etkin Bazı Yolaklar Üzerine PKC İzoenzim Aktivitelerinin Etkisinin in vitro İncelenmesi, Finished
The Scientific and Technological Research Council of Turkey - TUBITAK - RD : Ovaryum Kanser Hücrelerinden Salınan Eksozomların Primer Ovaryum Kanser Hücreleri İle Sağlıklı Epitel Ve Mezotel Hücreler Tarafından Alınım Yolları Ve Hedef Hücrelerdeki Karsinojenez Mekanizmasına Etkisi, Finished
The Scientific and Technological Research Council of Turkey - TUBITAK - RD : TCA Döngüsü ve Glikoliz Yolağındaki Metabolit Seviyelerinin Farklı Aşamalardaki Kolon Kanseri Hücre Hatlarında HPLC Yöntemi ile İncelenmesi, Finished
Contact

Ayar-Kayali Lab. on Biopharmaceutic Technologies and Bioanalysis
Research Group Leader
Hülya AYAR KAYALI
hulya.kayali@ibg.edu.tr
+90 232 299 41 00
(5081)
+9
0 232 299 41 58